Chapter 1: Physical Quantities, Units and Measurement
Objectives
Candidates should be able to:
- show understanding that all physical quantities consist of a numerical magnitude and a unit
- recall the following base quantities and their units: mass (kg), length (m), time (s), current (A), temperature (K), amount of substance (mol)
- use the following prefixes and their symbols to indicate decimal sub-multiples and multiples of the SI units: nano (n), micro (µ), milli (m), centi (c), deci (d), kilo (k), mega (M), giga (G)
- show an understanding of the orders of magnitude of the sizes of common objects ranging from a typical atom to the Earth
- state what is meant by scalar and vector quantities and give common examples of each
- add two vectors to determine a resultant by a graphical method
- describe how to measure a variety of lengths with appropriate accuracy by means of tapes, rules, micrometers and calipers, using a vernier scale as necessary
- describe how to measure a short interval of time including the period of a simple pendulum with appropriate accuracy using stopwatches or appropriate instruments
1.1 Physical Quantities and SI Units
- Physical quantities consist of:
- Numerical magnitude – denotes the size of the physical quantity.
- Unit – denotes the physical quantity it is expressing.
- Physical quantities can be classified into:
- Basic quantities
Basic Quantity Name of SI Unit SI Unit length metre m mass kilogram kg time second s thermodynamic temperature kelvin K amount of substance mole mol - Derived quantities – defined in terms of the basic quantities through equations. SI units for these quantities are obtained from the basic SI units through the equations.
Example 1.1
- Basic quantities

- Units of measurements: SI units are used as standardised units in all measurements in the world. SI is the short form for “International System of Units”.
- Other Units:
Length Mass Time 1 km = 1000 m 1 kg = 1000 g 1 h = 60 min 1 m = 100 cm 1 g = 1000 mg 1 min = 60 s 1 cm = 10 mm
- Examples of some derived quantities and their units:
Derived Quantity SI Unit area m2 volume m3 density kg/m3 speed m/s A complete list of key quantities, symbols and units used for the O Level examination can be found in the syllabus.
1.2 Prefixes, Symbols and Orders of Magnitude
- Physical quantities can be very large, like 23 150 000 000 m, or very small, like 0.000 000 756 m. Writing down such numbers can be time consuming and error-prone. We use prefixes to indicate decimal sub-multiples and multiples of the SI units to make writing such numbers easier.
- Some prefixes of the SI units are as follows:
Prefix Multiple Symbol Factor Order of Magnitude Tera 1 000 000 000 000 T 1012 12 Giga 1 000 000 000 G 109 9 Mega 1 000 000 M 106 6 Kilo 1000 K 103 3 Deci 0.1 d 10-1 -1 Centi 0.01 c 10-2 -2 Milli 0.001 m 10-3 -3 Micro 0.000 001 μ 10-6 -6 Nano 0.000 000 001 n 10-9 -9 Pico 0.000 000 000 001 p 10-12 -12 The ones in bold are specifically required in the syllabus.
Example 1.2- 0.000 0031 m = 3.1 μm = 3.1 * 10-6m
- 0.000 000 0012 s = 1.2 ns = 1.2 * 10-9s
- When measurements are too large or too small, it is convenient to express them in standard form as follows:
M × 10N
M lies in the range of: 1 ⩽ M < 10
N denotes the order of magnitude and is an integer. - Orders of magnitude are often being used to estimate numbers which are extremely large to the nearest power of ten.
E.g.- Estimate the number of strands of hair on a person’s head.
- Estimate the number of breaths of an average person in his lifetime.
- The following tables show how the orders of magnitude are used to compare some masses and lengths.
Mass/kg Factor Electron 10-30 Proton 10-27 Ant 10-3 Human 101 Earth 1024 Sun 1030 Length/m Factor Radius of a proton 10-15 Radius of an atom 10-10 Height of an ant 10-3 Height of a human 100 (100 = 1) Radius of the Earth 107 Radius of the Sun 109 Example 1.3
Find the ratio of the height of a human to that of an ant.
Ratio of height of human to that of an ant = 100/10-3 = 103 = 1000.
1.3 Scalars and Vectors
- A scalar quantity – has only magnitude but does not have direction.
E.g. mass, distance, time, speed, work, power. - A vector quantity – has both magnitude and direction.
E.g. weight, displacement, velocity, acceleration, force.
Example 1.4
The velocity of a particle can be stated as: “speed of particle = 2.0 m/s and it is moving at an angle of 30° above the horizontal”.

1.4 Addition of Vectors
1. Involves magnitude and direction.
Example 1.5
Find the resultant force R at point P due to F1 and F2.

Method 1: Trigonometric Method

Method 2: Graphical Method

(Not drawn to scale)
Step 1: Select an appropriate scale
E.g. 1 cm to 2 N.
Step 2: Draw a parallelogram of vectors to scale.
Step 3: Measure the diagonal to find R.
Step 4: Use the protractor to measure angle θ.
1.5 Measurement of Length
- Choice of instrument depends on the degree of accuracy required.
Range of length, l Instrument Accuracy Example l > 100 cm Measuring tape ± 0.1 cm waistline of a person 5 cm < l < 100 cm Metre rule ± 0.1 cm height of an object 1 cm < l < 10 cm Vernier calipers ± 0.01 cm diameter of a beaker l < 2 cm Micrometer screw gauge ± 0.001 cm thickness of a length of wire - How parallax errors can occur during measurement:
- incorrect positioning of the eye

- the object is not touching the marking of the scale (for measuring tape and metre rule, ensure that the object is in contact with the scale)

- A measuring instrument can give precise but not accurate measurements, accurate but not precise measurements or neither precise nor accurate measurements.
- Precision is how close the measured values are to each other but they may not necessarily cluster about the true value. Zero errors and parallax errors affect the precision of an instrument.
- Accuracy is how close a reading is to the true value of the measurement. The accuracy of a reading can be improved by repeating the measurements.
- Vernier calipers
A pair of vernier calipers can be used to measure the thickness of solids and the external diameter of an object by using the external jaws. The internal jaws of the caliper are used to measure the internal diameter of an object. The tail of the caliper is used to measure the depth of an object or a hole. Vernier calipers can measure up to a precision of ±0.01 cm.

- Precautions: Check for zero error and make the necessary correction.
Example 1.6- Positive zero error:

- Zero error = +0.02 cm
- Negative zero error:

- Zero error = –0.02 cm
Note: In (b), the pair of vernier calipers is built with an existing zero error. There is a negative reading without any object between its jaws. The vernier scale is pushed 0.02 cm to the left.
Example 1.7
When the jaws of a pair of vernier calipers are closed, the vernier caliper reading is as shown.
- Zero error = –0.02 cm

- When the same pair of vernier calipers is used to measure the diameter of a beaker, the vernier caliper reading is as shown.

- What is the diameter of the beaker?
Solution
Zero Error = +0.01 cm
Reading = 4.00 + 0.01 = 4.01 cm
Actual reading = 4.01 − 0.01 = 4.00 cm
- What is the diameter of the beaker?
- Micrometer screw gauge

- Precautions:
- Ensure that the jaws of the micrometer screw gauge are completely closed by turning the ratchet until you hear a ‘click’ sound.
- Check that the ‘0’ mark of the thimble scale is completely in line with the horizontal line of the reference scale. If not, there is zero error.
- Example 1.8
- Positive zero error: ‘0’ mark is below the horizontal line

- Zero error = +0.06 mm
- Negative zero error: ‘0’ mark is above the horizontal line

- Zero error = –0.04 mm
- Example 1.9
A micrometer screw gauge is used to measure the thickness of a plastic board. When the jaws are closed without the plastic board in between, the micrometer reading is shown in Fig. (a).

- With the jaws closed around the plastic board, the micrometer reading is shown in Fig. (b).

- What is the thickness of the plastic board?
Solution
Zero error = +0.03 mm
Reading = 13.5 + 0.49 = 13.99 mm
Actual thickness of plastic board = 13.99 – (+0.03) = 13.96 mm
Example 1.10
The micrometer reading as shown in the figure is 12.84 mm.

- What is the value of x on the circular scale?
Solution
Reading = 12.5 + reading on the circular scale = 12.84 mm
Reading on the circular scale = 12.84 – 12.5 = 0.34 mm
Since the marking x is 1 mark above 0.34 mm, the value of x is 35.
- Period of oscillation of a simple pendulum.

- One oscillation – One complete to-and-fro movement of the bob from point A to B to C and back to A.
- Period, T – Time taken for one complete oscillation.
- Amplitude – The distance between the rest position of the bob (point B) to the extreme end of the oscillation (either point A or point C).
- Steps to find the period of oscillation:
- Step 1: Take the total time for 20 oscillations.
- Step 2: Repeat Step 1.
- Step 3: Take the average of the two timings.
- Step 4: Divide the average in Step 3 by 20 to obtain the period.
- The period of the pendulum, T, is affected only by its length, l, and the acceleration due to gravity, g.
t = 2π √(T/g)
T is not affected by the mass of the pendulum bob.
- Example 1.11
A pendulum swings backwards from B to A and forwards to C passing through B, he middle point of the oscillation. The first time the pendulum passes through B, a stopwatch is started. The thirtieth-time the pendulum passes through B, the stopwatch is stopped and the reading taken is 25.4 seconds. What is the period of the pendulum?
Solution

Chapter 2: Kinematics
Objectives
Candidates should be able to:
- state what is meant by speed and velocity
- calculate average speed using distance travelled / time taken
- state what is meant by uniform acceleration and calculate the value of an acceleration using change in velocity / time taken
- interpret given examples of non-uniform acceleration
- plot and interpret a displacement-time graph and a velocity-time graph
- deduce from the shape of a displacement-time graph when a body is:
- at rest
- moving with uniform velocity
- moving with non-uniform velocity
- deduce from the shape of a velocity-time graph when a body is:
- at rest
- moving with uniform velocity
- moving with uniform acceleration
- moving with non-uniform acceleration
- calculate the area under a velocity-time graph to determine the displacement travelled for motion with uniform velocity or uniform acceleration
- state that the acceleration of free fall for a body near to the Earth is constant and is approximately 10 m/s2
- describe the motion of bodies with constant weight falling with or without air resistance, including reference to terminal velocity
2.1 Distance vs Displacement and Speed vs Velocity
Scalar Vector Distance Displacement Speed Velocity
- Example 2.1
A car travelled from point O to D along the curved path OABCD.

- The distance travelled by the car is OABCD.
The displacement of the car from point O is OD (to the right of O).
- When measuring/ calculating the displacement of an object, one has to include its starting point.
Example 2.2
Wrong: “The displacement of the bus is 500 m.” (500 m from where?)
Right: “The displacement of the bus from point A is 500 m in the backward direction.” or “The displacement of the bus from point A is –500 m (taking the forward direction as positive).” - The formula for calculating speed is
Speed = Distance travelled/Time taken - Average speed = Total distance travelled/Total time taken
- Velocity is the rate of change of displacement of an object from a fixed point (displacement per unit time).
- Average velocity = Resultant displacement from a fixed point/Total time taken
The average velocity vavg of an object moving through a displacement (Δx) along a straight line in a given time (Δt) is:
- The formula for calculating speed is

2.2 Acceleration
- Acceleration is the rate of change of velocity.
- a = Δx/Δt = (v–u)/Δt = where v is the final velocity, u is the initial velocity and Δt is the time taken.
- Acceleration is a vector quantity. (You need to give both the magnitude and direction when writing down the answer.)
2.3 Graph of Distance vs Time
- The distance-time graph of a moving object along a straight road is used to find its speed.
- The gradient of the graph gives the speed of the object.
Example 2.3
Object moving at uniform speed

2.4 Graph of Speed vs Time
- The speed-time graph of a moving object along a straight road is used to find:
- Acceleration (Using the gradient of graph)
- Distance travelled (Using the area under the graph)
Example 2.4
Object moving with uniform acceleration:

- For an object moving with constant acceleration, the speed-time graph is a sloping straight line. A constant acceleration means that speed is increasing at a constant rate.
2.5 Interpret Other Speed-Time Graphs (Non-Uniform Acceleration)
- Increasing acceleration:

- Notice that the gradient of the graph becomes steeper.
The gradient of triangle 2 is larger than the gradient of triangle 1.
(Gradient gets more and more positive).
The speed is increasing with increasing acceleration (increasing rate).
At time = t1, acceleration = a1.
At time = t2, acceleration = a2.
a2 > a1
- Decreasing acceleration:

- Notice that the gradient of the graph becomes less steep.
The gradient of triangle 2 is smaller than the gradient of triangle 1.
(Gradient gets less and less positive).
The speed is increasing with decreasing acceleration (decreasing rate).
At time = t1, acceleration = a1.
At time = t2, acceleration = a2.
a2 < a1
2.6 Acceleration Due to Free-Fall
Near the surface of the earth, the acceleration of free fall for an object is constant and is approximately 10 m/s2. When an object drops from the top of a building, its speed will increase from 0 m/s uniformly at a rate of 10 m/s per second.
2.7 Effect of Air Resistance
- In real life, a falling object will encounter air resistance on Earth, unless it is moving in a vacuum.
- Air resistance acts against the motion of the object increasingly to reduce its downward acceleration (NOT SPEED) to zero.
- When the air resistance increases till it is equal to the weight of a falling object, the acceleration of the object is zero.
- With zero acceleration, the object will continue falling downward at a constant velocity.
- The constant velocity of the object is known as “terminal velocity”.
Example 2.5
An astronaut standing on the Moon’s surface throws a rock vertically upwards. The figure shows the speed-time graph of the rock where at t = 0 s, the rock just leaves the astronaut’s hand. Air resistance on the Moon can be neglected.

- What is the time taken for the rock to reach its maximum height?
- What is the total distance travelled by the rock when it returns to its initial position?
- Find the acceleration of the rock.
- The rock is then brought back to the Earth’s surface and the astronaut repeats the same action as on the Moon. Determine whether the speed- time graph of the rock, when it is thrown on Earth, will be different. Explain your answer.
Solution
- From the graph, the time taken for the rock to reach its maximum height is 3.90 seconds.
- Total distance travelled = 2 × area under the graph
= {½×(6.50 − o.oo)×(3.90 − o.00)}×2
= 25.4m (to 3 s.f.) - Acceleration of rock = (6.50 − 0.00)/(0.00 − 3.90)
= −1.67 m s2 or −1.67 m s−2 (to 3 s.f.)
- The speed-time graph of the rock on Earth is different because the speed of the rock decreases as it falls from a height. This is due to air resistance. The speed of the rock is decreasing at an increasing rate. The deceleration of the rock increases as the speed decreases. Hence, the gradient of the speed-time graph is steeper initially and becomes gentler after some time. The sketch of the speed-time graph is a curve and not a straight line.
Example 2.6
The graph shows the speed of a car from the time the driver saw an obstacle on the road and applied the brakes till the car came to a stop.

- How long did it take the driver to begin applying the brakes after seeing the obstacle?
- Calculate the distance travelled
- before the brakes were applied,
- while the brakes were being applied.
- Calculate the average speed of the car.
Solution
- The speed remains at 20 m/s for the first 0.5 seconds, so the driver took 0.5 seconds to begin applying the brakes after seeing the obstacle.
- Distance travelled before braking
= 20 × 0.5
= 10m - Distance travelled while the brakes were being applied
= ½ × 20 × (4.5 – 0.5)
= 40m
- Distance travelled before braking
- Average speed of car = (Total distance travelled)/(Total time taken)
= (10 + 40)/4.5
= 50/4.5
= 11.1 m/s or 11.1 m s-1 (to 3 s.f)
Chapter 3: Dynamics
Objectives
Candidates should be able to:
- apply Newton’s Laws to:
- describe the effect of balanced and unbalanced forces on a body
- describe the ways in which a force may change the motion of a body
- identify action-reaction pairs acting on two interacting bodies (stating of Newton’s Laws is not required)
- identify forces acting on an object and draw free body diagram(s) representing the forces acting on the object (for cases involving forces acting in at most 2 dimensions)
- solve problems for a static point mass under the action of 3 forces for 2-dimensional cases (a graphical method would suffice)
- recall and apply the relationship resultant force = mass × acceleration to new situations or to solve related problems
- explain the effects of friction on the motion of a body
3.1 Forces
- A force (SI unit: Newton, symbol: N) is a push or a pull exerted on a body by another body, i.e. an object resting on a table will have a contact force (normal force) acting on it upwards. This force is equal to its weight.

- Effects of a force on a body:
- Increase/ decrease speed of a body (accelerate/ decelerate)
- Change direction of a moving body
- Newton’s First Law:
A body will remain stationary or in continuous linear motion unless acted upon by a resultant force. - Newton’s Second Law:
Resultant vector sum of forces on body is given by:
F = ma
where m is the mass of the body and a is the acceleration of the body in the direction of F. - Newton’s Third Law:
For every action, there is an equal and opposite reaction.
3.2 Balanced and Unbalanced Forces
- Balanced forces: If resultant F = 0 N, the body is either stationary or moving with constant velocity.
Example 3.1
A parachutist falls to the ground at terminal velocity when his weight is equal to the upward force acting on him due to air resistance. Hence, the resultant force acting on him is zero, i.e. his acceleration is zero. - Unbalanced forces: If resultant F ≠ 0 N,
- a stationary body will start moving,
- a moving body will change its velocity.
3.3 Friction
- Friction is the force which opposes motion when objects slide over each other. For a moving object, the friction on the object acts in the direction opposite to its motion.
- Advantages of friction:
- Walking on roads.
- Friction in brake pads and wheels of cars and bicycles.
- Disadvantages of friction:
- Wears down moving parts of machines.
- For an object moving on a rough surface, more energy is needed to move the object as compared to moving on a smooth surface.
- For an object moving on a rough surface, energy is required for the object to maintain a constant speed. Otherwise, it will slow down and come to a stop.
- Ways to overcome friction:
- Use lubricant (i.e. graphite or oil) for moving parts of machines.
- Use ball-bearings between moving surfaces.
- Make sure moving parts of machines have very smooth surfaces.
Example 3.2
An object weighing 50 N lies on a rough surface. A constant F1 force of 12 N acts on the object. The frictional force f acting on the object is 2 N. Find the acceleration of the object. (Take acceleration due to gravity to be 10 m/s2.)

Solution
Vertically, resultant force = normal force – weight = 0 N
Horizontally, resultant force R = F1 – f = 12 – 2 = 10 N
(Object will only accelerate on horizontal plane)
Mass of object = 50/10 Kg
= 5Kg
Using formula:
F = ma
10 = 5a
a = 2 m/s2
(Object is accelerating at 2 m/s2 to the right, i.e. in the direction of F.)
Example 3.3
An object moves in a circular path at a constant speed. Is the object accelerating?
Solution
Yes. Its velocity keeps changing (because direction keeps changing), hence there is a resultant force causing the change. Resultant force acts towards the centre of the circle.
Example 3.4
A skydiver of mass 60 kg falls from rest vertically downwards at a constant velocity.
The figure shows the forces, P and Q, acting on him.

- Identify the forces P and Q acting on the skydiver.
- Explain why P is acting upwards.
- When the skydiver starts to fall from rest, the forces P and Q are unbalanced.
- Find P and Q at t = 0 s.
- Find P and Q when the velocity of the skydiver is uniform.
- Describe, in terms of the forces acting on the sky diver, why the velocity of the skydiver increases before reaching terminal velocity.
Solution
- P is the air resistance on the skydiver and Q is the weight of the skydiver.
- Air resistance opposes the motion of the skydiver. Since the skydiver is falling vertically downwards, the air resistance acting on him is in the upward direction to oppose his motion.
- Take all forces acting downwards as positive.
- P = 0 N
Q = mg = 60 × 10 = 600 N - When the velocity of the skydiver is uniform, he has reached terminal velocity. The resultant force acting on him is 0 N.
Q – P = 0
P = Q = 600 N - As a result of unbalanced forces, there will be a non-zero resultant force acting on the skydiver, and it is acting vertically downwards. By Newton’s 2nd Law, the skydiver is accelerating downwards. Hence, the velocity of the skydiver increases before it reaches terminal velocity.
- P = 0 N
Chapter 4: Mass, Weight and Density
Objectives
Candidates should be able to:
- state that mass is a measure of the amount of substance in a body
- state that mass of a body resists a change in the state of rest or motion of the body (inertia)
- state that a gravitational field is a region in which a mass experiences a force due to gravitational attraction
- define gravitational field strength, g, as gravitational force per unit mass
- recall and apply the relationship weight = mass × gravitational field strength to new situations or to solve related problems
- distinguish between mass and weight
- recall and apply the relationship density = mass / volume to new situations or to solve related problems
4.1 Mass
- Defined as a measure of the amount of substance in a body. (SI unit: kilogram, symbol: kg)
- The magnitude of mass depends on the size of the body and the number of atoms in the body.
- Mass is a scalar quantity.
4.2 Inertia
- Defined as the resistance of the body to change in its state of rest or motion due to its mass.
- To overcome inertia of a body, a force has to be applied. This force is dependent on the body’s mass.
4.3 Gravitational Field Strength
Defined as the gravitational force acting on a body per unit mass.
| Gravitational Field Strength | |
| Earth | 10 N kg-1 |
| Moon | 1.6 N kg-1 |
Note: These are approximate values for points close to and on the planets’ surface.
i.e. on Earth, a force of 10 N is pulling on a 1 kg falling object.

Note: Since the resultant force on object is 10 N (weight), the acceleration of the object is (by Newton’s 2nd Law) 10 m s–2.
4.4 Weight
- Defined as the gravitational force W acting on an object of mass m.
- When a body falls, its gravitational force (weight) can produce an acceleration, g (the acceleration due to gravity).
- Using Newton’s 2nd Law of F = ma, we have W = mg.
- Comparison of weight and mass:
| Mass | Weight | ||
| 1. | definition | the amount of substance in a body | the gravitational pull acting on a body |
| 2. | depends on location | no | yes |
| 3. | measured by using | beam balance | spring balance |
| 4. | unit | kilogram | Newton |
4.5 Density
- The density of a body, ρ, is defined as its mass, m, per unit volume, V.
Density = Mass/Volume
ρ = m/V
- SI Unit: kg m–3
- For an object to float in a liquid, the object has to be less dense than the liquid. As such, if an object is denser than the liquid, the object will sink in the liquid.

Chapter 5: Turning Effect of Forces
Objectives
Candidates should be able to:
- describe the moment of a force in terms of its turning effect and relate this to everyday examples
- recall and apply the relationship moment of a force (or torque) = force × perpendicular distance from the pivot to new situations or to solve related problems
- state the principle of moments for a body in equilibrium
- apply the principle of moments to new situations or to solve related problems
- show understanding that the weight of a body may be taken as acting at a single point known as its centre of gravity
- describe qualitatively the effect of the position of the centre of gravity on the stability of objects
5.1 Moment of a Force
- Moment – the turning effect of a force about a pivoting point
- Moment of force = F × d
F: Force
d: perpendicular distance of line of action of F from pivot

- SI unit of moment: N m
- Conditions for object to be in equilibrium:
- The sum of moments about any point is zero. (Principle of Moments)
- The vector sum of forces on object is zero.
- Principle of Moments:
For an object in equilibrium, the sum of clockwise moments about any point is equal to the sum of anticlockwise moments about the same point. (Resultant moment = 0 N m)
5.2 Centre of Gravity (C.G.)
- The C.G. of an object is the point where the whole weight appears to act on.
- The C.G. will not change regardless of how the object is orientated.
- The C.G. can lie outside an object.
E.g. C.G. of a metal ring is in the middle of the circle.

5.3 Stability
- Stability – the ability of an object to retain its original position after being displaced slightly.

- The stability of an object can be improved by:
- Lowering its C.G. (Add weights to the object’s lower part).
- Increasing the base area of the object.
Chapter 6: Pressure
Objectives
Candidates should be able to:
- define the term pressure in terms of force and area
- recall and apply the relationship pressure = force / area to new situations or to solve related problems
- describe and explain the transmission of pressure in hydraulic systems with particular reference to the hydraulic press
- recall and apply the relationship pressure due to a liquid column = height of column × density of the liquid × gravitational field strength to new situations or to solve related problems
- describe how the height of a liquid column may be used to measure the atmospheric pressure
- describe the use of a manometer in the measurement of pressure difference
6.1 Pressure
- Pressure is the force acting per unit area.
Pressure = Force/Area
SI unit: Pascal (Pa) or N m–2
6.2 Liquid Pressure
- An object immersed in a uniform liquid will experience a pressure which depends only on the height of the liquid above the object.

- Pressure at point A due to the liquid, P = ρgh

- When a liquid is at equilibrium, the pressure is the same at any point along the same horizontal surface. Thus the liquid in the container settles at a common height, h.

6.3 Transmission of Pressure in Hydraulic System
- Pressure can be transmitted in all directions if it is exerted on an incompressible fluid in a container.
- Components of a hydraulic system:
- Container with two openings
- A press
- A piston
- Incompressible fluid
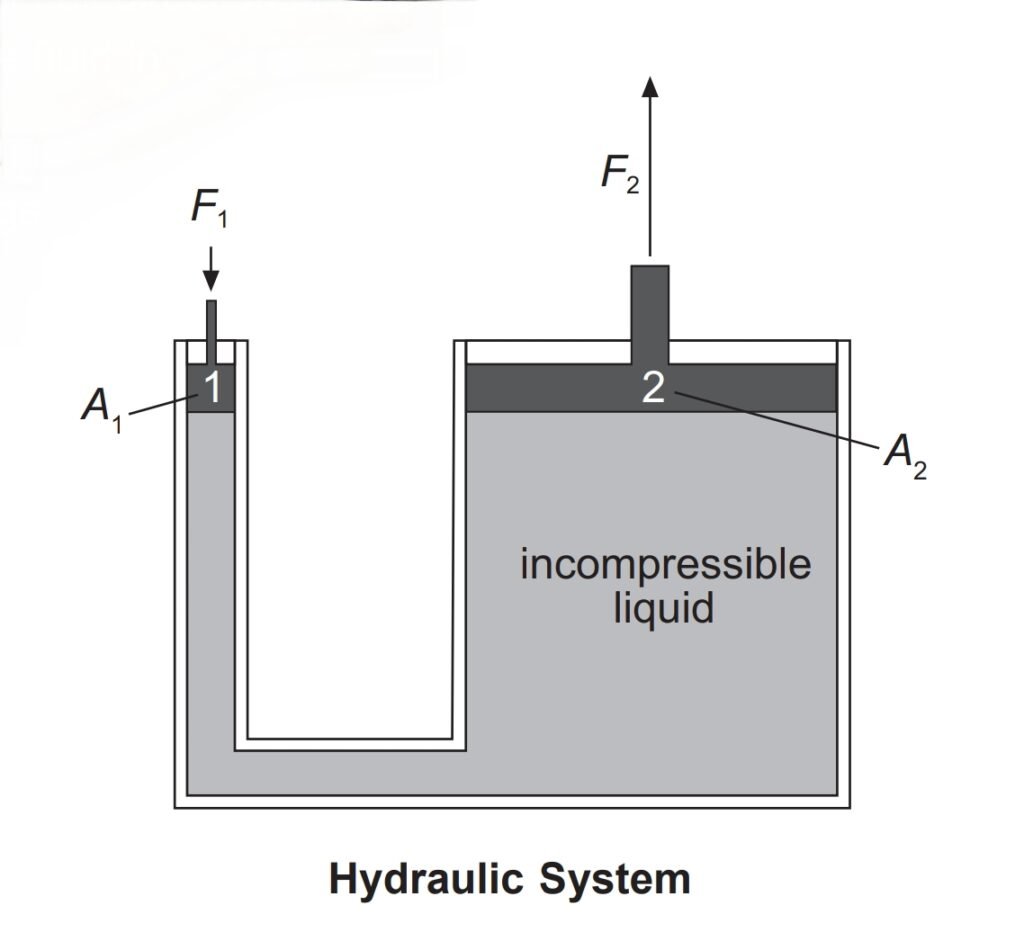
- In the above figure, if the two pistons at ‘1’ and ‘2’ have the same area, then the force F1 exerted on one piston will have the same magnitude as F2 at the other piston.
- If the area A1 is smaller than area A2, then the force exerted at ‘1’ will produce a larger force at ‘2’.
Pressure = P = F1/A1 = F2/A2 - Thus we can use a hydraulic system to lift heavy objects.
6.4 Atmospheric Pressure
- Defined as the force per unit area exerted against a surface by the weight of air above that surface.
- Instrument to measure atmospheric pressure: mercury barometer
- At sea-level, h = 760 mm.
Atmospheric pressure recorded as 760 mm Hg. - Even if the tube is tilted, h will still remain the same unless it is brought to a different level where the atmospheric pressure is different.

6.5 Manometer
- The manometer is an instrument that is used to measure gas pressure.
- Gas pressure, P1 = P0 + ρgh where h – difference in height.

Chapter 7: Energy, Work and Power
Objectives
Candidates should be able to:
- show understanding that kinetic energy, potential energy (chemical, gravitational, elastic), light energy, thermal energy, electrical energy and nuclear energy are examples of different forms of energy
- state the principle of conservation of energy and apply the principle to new situations or to solve related problems
- calculate the efficiency of an energy conversion using the formula efficiency = energy converted to useful output / total energy input
- state that kinetic energy Ek = ½ mv2 and gravitational potential energy Ep = mgh (for potential energy changes near the Earth’s surface)
- apply the relationships for kinetic energy and potential energy to new situations or to solve related problems
- recall and apply the relationship work done = force × distance moved in the direction of the force to new situations or to solve related problems
- recall and apply the relationship power = work done / time taken to new situations or to solve related problems
7.1 Energy
- Different forms: kinetic energy (KE), elastic potential energy, gravitational potential energy (GPE), chemical potential energy, thermal energy.
- SI unit: Joule (J)
- Principle of Conservation of Energy: The total energy in a system remains constant and cannot be created or destroyed. It can only be converted from one form to another without any loss in the total energy.
7.2 Gravitational Potential Energy (GPE) and Kinetic Energy (KE)
- Take the surface of the Earth to be the reference level (GPE = 0).
GPE of an object of mass m at height h above surface:
GPE = mgh - KE of a moving object of mass m, with a velocity v is
KE = ½ mv2
Example 7.1
For a free-falling object of mass m, its gravitational potential energy is converted into kinetic energy. Take ground level as reference level (GPE = 0).

Apply the Principle of Conservation of Energy and assuming there is no air resistance:
Total energy at height h = Total energy at ground level
mgh = ½ mv2
Velocity of object, v = √2gh
Note: The total energy of the object is constant throughout its fall, not just at the two positions used in the above calculation.
(GPE + KE = Total energy = Constant)
7.3 Work
- Energy is required for an object to do work.
- Defined as the product of applied force (F) and the distance moved (s) in the direction of the force.
W = Fs (Unit: J)
No work is done if the applied force F does not displace the object along the direction of the force.
7.4 Power
- Defined as the rate of work done.
Power = Work done/Time taken
SI unit: Watt (W) or J s–1
Efficiency of an energy/ power conversion:

Example 7.2
A box with a mass of 30 kg can be lifted by a light rope threaded through a smooth pulley.

- If the box is lifted at a constant speed from the ground to a height of 2.0 m in 4.0 s, what is the power required?
- If the box is lifted with a constant acceleration of 1.5 m/s2 from rest to a height of 3.0 m above the floor, what is the power required?
Take g, the gravitational field strength as 10 N/kg.
Solution
- Draw a free body diagram of the box and identify all the forces acting on it.

Take forces acting upwards to be positive.
Using Newton’s 2nd Law,
F – mg = 0
∴ F = mg
= (30)(10)
= 300 N
Power required = rate of work done
= Fs/t
= (300 × 2.0)/4.0
= 150 W

Take forces acting upwards to be positive.
Using Newton’s 2nd Law,
F – mg = ma
F – 300 = 30 × 1.5
F = 45 + 300
F = 345 N
Sketch the speed-time graph of the box to obtain the time taken for the box to move to a height of 3.0 m above the ground.

From the graph, we can obtain the velocity (gradient of graph) and the total displacement of the box.
Gradient of velocity-time graph,
a = (v-u)/t = (v-0)/t
1.5 = v/t
v = 1.5t ——-(1)
Area under the graph (shaded) = Displacement s of box from the ground
s = ½ × Base × Height = ½ × t × v
s = ½ vt
½ vt = 3.0 ——-(2)
Substitute (1) into (2):
½ (1.5t)t = 3.0
¾ t2 = 3.0
t2 = 4.0
(t – 2.0)(t + 2.0) = 0
t = 2.0 s(since t > 0)
Power required = (345 × 3.0)/2.0
= 518 W (to 3 s.f)
Chapter 8: Kinetic Model of Matter
Objectives
- compare the properties of solids, liquids and gases
- describe qualitatively the molecular structure of solids, liquids and gases, relating their properties to the forces and distances between molecules and to the motion of the molecules
- infer from Brownian motion experiment the evidence for the movement of molecules
- describe the relationship between the motion of molecules and temperature
- explain the pressure of a gas in terms of the motion of its molecules
- recall and explain the following relationships using the kinetic model (stating of the corresponding gas laws is not required):
- a change in pressure of a fixed mass of gas at constant volume is caused by a change in temperature of the gas
- a change in volume occupied by a fixed mass of gas at constant pressure is caused by a change in temperature of the gas
- a change in pressure of a fixed mass of gas at constant temperature is caused by a change in volume of the gas
- use the relationships in (f) in related situations and to solve problems (a qualitative treatment would suffice)
8.1 States of Matter
The 3 States of Matter
| Solid | Liquid | Gas | |
| Volume | Definite | Definite | Indefinite (Takes the shape and size of container) |
| Shape | Definite | Indefinite (Takes the shape of container) | Indefinite (Takes the shape of container) |
| Compressibility | Not compressible | Not compressible | Compressible |
| Arrangement of atoms/molecules |
|
|
|
| Density | High (Usually) | High | Low |
| Forces between atoms/ molecules | Very strong | Strong | Very Weak |
| Movement of atoms/ molecules | Can only vibrate about fixed positions | Able to move pass each other and not confined to fixed positions | Move in random manner independent of each other and at high speed. |
Common mistakes:
- Some substances, such as carbon dioxide, are commonly known to be in gaseous state at room temperature. However, this does not mean that the carbon dioxide molecules move in random motion.
(Check its state (temperature): solid or gas, etc.)
E.g. Dry ice is a solid which consists of carbon dioxide molecules in an orderly arrangement. - Not all solids have high density, i.e. “ice” is a solid consisting of water molecules arranged orderly in an open hollow structure. Hence, its density is lower than water (liquid) and it can float in water.
8.2 Brownian Motion
The random and irregular motion of gas and liquid molecules. Experimental observation (using microscope): Smoke particles in a sealed glass cell move about randomly and irregularly, because of bombardment by air molecules in the cell.

8.3 Pressure of Gas
- In a sealed container, gas can exert pressure on the walls of the container.
- The large number of molecules move at high speed, colliding against the container’s walls and exerting a force against the wall when they bounce off the walls.
- The force per unit area exerted by the molecules on the wall is the pressure of the gas on the wall.
- Gas pressure increases when the
- number of molecules in the container increases,
- speed of molecules increases,
- molecules have larger mass.
8.4 Relationship between Pressure (P), Volume (V) and Temperature (T)
For a constant mass of gas:

Example 8.1
To get a linear graph that shows P is inversely proportional to V, rearrange the equation:
P1V1 = P2V2 = constant, k
P1 = k/V1
Sketch the graph of P against 1/V :
y-axis(P), x-axis (1/V), gradient = k

Chapter 9: Transfer of Thermal Energy
Objectives
Candidates should be able to:
- show understanding that thermal energy is transferred from a region of higher temperature to a region of lower temperature
- describe, in molecular terms, how energy transfer occurs in solids
- describe, in terms of density changes, convection in fluids
- explain that energy transfer of a body by radiation does not require a material medium and the rate of energy transfer is affected by:
- colour and texture of the surface
- surface temperature
- surface area
- apply the concept of thermal energy transfer to everyday applications
9.1 Types of Heat Transfer
- types of heat transfer: Conduction, Convection, Radiation
- Transfer of thermal energy is always from a high temperature region to a low temperature region (Temperature gradient).
| Conduction | Convection | Radiation | |
| Medium | Solids Liquids Gases | Liquids (fluid) Gases (fluid) | Vacuum* |
| Process |
| Movement of atoms/ molecules in the form of convection by currents set up by density change in parts of the fluid being heated. | Infrared waves (no medium required) |
- Radiation does not require matter to transfer heat, but radiation can travel through matter (through several thousands of metres in air or a few metres in common solids).
9.2 Conduction
- A direct contact between media is necessary.
- Metals are the best solid conductors because of their free electrons.
- Liquids and gases are poor conductors because their molecules are not closely packed together in fixed positions like solids.
- Application: Use metals to make cooking utensils.
9.3 Convection
- Molecules/ atoms must be free to move.
- Set-up of a convection current: The fluid closer to the heat source expands, and its density decreases and the surrounding denser fluid displaces it.
- Application: Air conditioners are placed near the ceiling because cold air, being denser, will sink to displace the warm air in the room.
9.4 Radiation
- Factors affecting radiation:
- Colour
- Roughness
- Area exposed to radiation
- Good radiator/ good absorber of radiation: black, dull surface, with a huge amount of surface area exposed.
- Poor radiator/ poor absorber of radiation: bright, shiny and polished surface.
- Application: Greenhouses for growing plants.
9.5 Vacuum Flask
- Reduces heat transfer in or out through conduction, convection and radiation.
- Can store and maintain temperature (either hot or cold) of the contents in the flask.
| Type of heat transfer | How heat transfer is reduced |
| Convection | Vacuum between the double glass walls. |
| Conduction | Vacuum between the double glass walls. Insulated cover and stopper. |
| Radiation | Shiny silvered inner surface of the glass walls. |
Chapter 10: Temperature
Objectives
Candidates should be able to:
- explain how a physical property which varies with temperature, such as volume of liquid column, resistance of metal wire and electromotive force (e.m.f.) produced by junctions formed with wires of two different metals, may be used to define temperature scales
- describe the process of calibration of a liquid-in-glass thermometer, including the need for fixed points such as the ice point and steam point
10.1 Temperature
- A measure of the degree of ‘hotness’ or ‘coldness’ of a body.
- SI Unit: Kelvin (K)
- Commonly-used unit is degree Celsius (°C): θ (K) = θ (°C) + 273.15
10.2 Measurement of Temperature
- Material for temperature measurement: Substance/ material which possesses temperature-dependent property and thus can change continuously with temperature variations.
Temperature-dependent (Thermometric) Properties:
| Thermometric Property | Thermometer | Range |
| Volume of a fixed mass of liquid (e.g. mercury or alcohol) | Mercury Alcohol Clinical thermometer | –10 ºC to 110 ºC – 60 ºC to 60 ºC 35 ºC to 42 ºC |
| Electromotive force (e.m.f.) (between hot and cold junctions of two different metals joined together) | Thermocouple | –200 ºC to 60 ºC Common ones |
| Resistance of metal e.g. Platinum | Resistance thermometer | –200 ºC to 1200 º C |
| Pressure of a fixed mass of gas at constant volume | Constant-volume gas thermometer | Estimated –258 ºC to 1027 ºC |
10.3 Temperature Scale
- Temperature is measured with reference to 2 fixed points:
- Lower Fixed Point or Ice point (0 °C):
Temperature of pure melting ice at standard atmospheric pressure. - Upper Fixed Point or Steam point (100 °C):
Temperature of pure boiling water at standard atmospheric pressure.
- Lower Fixed Point or Ice point (0 °C):
- The length between the 2 fixed points is divided into 100 equal intervals of 1 °C.
- Apply the following general formula to calculate temperature of a material:

- where:
θ is temperature of material
X0 is thermometric property at θ
X100 is thermometric property at steam point
X0 is thermometric property at ice point
i.e. for clinical thermometer, X is the length of the mercury thread at temperature θ; for thermocouples, it is the voltmeter reading at temperature θ.
10.4 The Thermocouple
- To measure the temperature of an unknown substance:
- One junction is kept at a constant temperature (i.e. ice point).
- The other junction is kept at the point where the temperature is to be measured.

- Advantages:
- Can withstand high temperature with suitable metals.
- Large temperature range. Can measure very low or very high temperatures.
- Junctions used are sharp and pointed and therefore can be used to measure temperature accurately at a point.
- Rapid response to temperature change.
Chapter 11: Thermal Properties of Matter
Objectives
Candidates should be able to:
- describe a rise in temperature of a body in terms of an increase in its internal energy (random thermal energy)
- define the terms heat capacity and specific heat capacity
- recall and apply the relationship thermal energy = mass × specific heat capacity × change in temperature to new situations or to solve related problems
- describe melting/ solidification and boiling/ condensation as processes of energy transfer without a change in temperature
- explain the difference between boiling and evaporation
- define the terms latent heat and specific latent heat
- recall and apply the relationship thermal energy = mass × specific latent heat to new situations or to solve related problems
- explain latent heat in terms of molecular behaviour
- sketch and interpret a cooling curve
11.1 Introduction
- Temperature – a measure of the internal energy of the substance’s atoms/molecules.
- Increase in temperature – caused by the supply of heat which increases internal energy.
- Internal energy – sum of kinetic energy and potential energy of the atoms/molecules.
- Types of internal energy:
| State of substance | Type of internal energy |
| Solid | vibrational kinetic energy + potential energy |
| Liquid | translational kinetic energy + potential energy |
| Gas | mainly translational kinetic energy |
11.2 Change of States
- Two main changes occur when heat is supplied to a substance
- Increase in temperature
- Change of state (i.e. solid to liquid)
- The following chart shows the changes of state (without temperature change) and their corresponding processes involved:

- + Thermal Energy: Heat is absorbed by substance
– Thermal Energy: Heat is removed from substance (Released to surroundings)
| 1. Melting |
|
| 2. Boiling |
|
| 3. Freezing |
|
| 4. Condensation |
|






