This lesson covers:
- Brownian motion
- Diffusion
- The effect of temperature on particle movement
Brownian Motion
Brownian motion describes the random, zigzag movement of tiny particles when suspended in a liquid or gas.
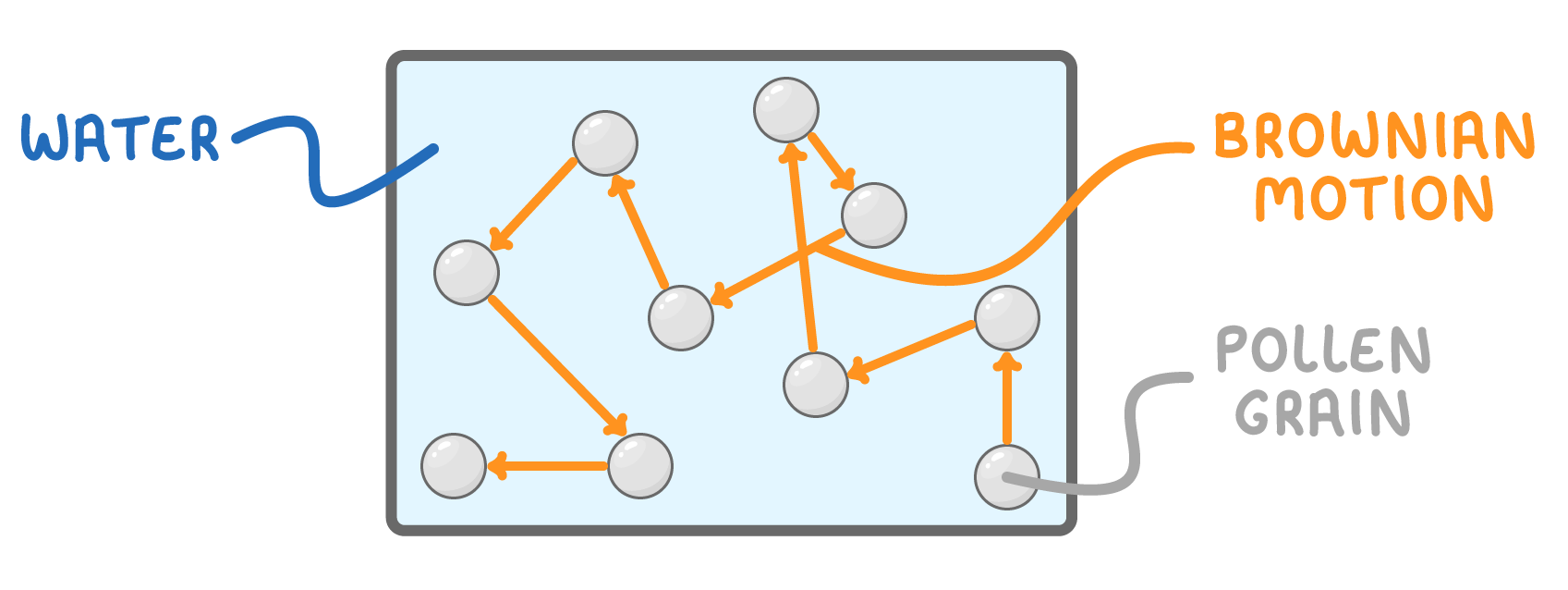
- In 1827, scientist Robert Brown observed the irregular motion of pollen particles suspended in water under a microscope.
- This type of random particle movement was later termed Brownian motion.
- Brownian motion involves lighter particles like air molecules colliding with heavier particles like smoke, causing the heavier particles to move around randomly.
Random motion of particles causes diffusion
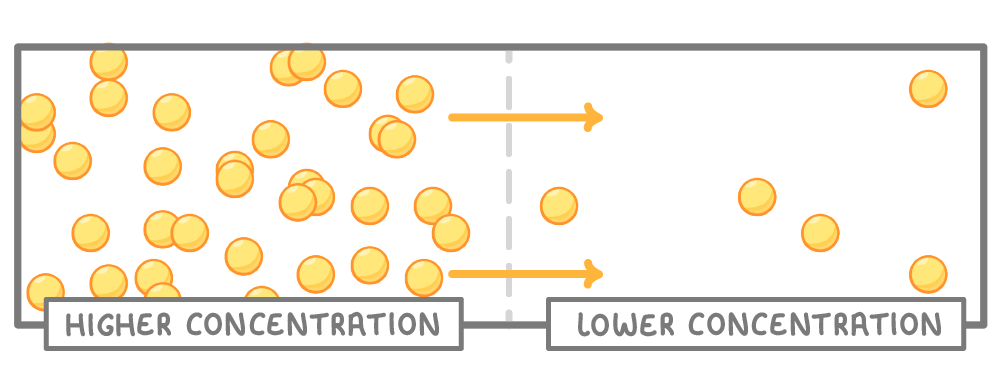
- Particles randomly move around and collide.
- This causes them to spread from areas of high concentration to areas of low concentration.
- This continues until particles are evenly dispersed throughout.
Increasing temperature, increases particle movement

- At higher temperatures, particles have more kinetic energy so move quicker and collide more forcefully.
- With faster, more energetic motion, particles require more space between each other. This causes expansion of the material.
- There is increased particle collision with container walls, increasing the pressure.
PRACTICE QUIZ
1. What is particle movement?
Answer: Particle movement refers to the motion of individual atoms, molecules, or particles in a substance. This motion can be random, as seen in gases and liquids, or more restricted, as in solids.
2. What is Brownian motion?
Answer: Brownian motion is the random, zigzag movement of tiny particles when suspended in a fluid (liquid or gas). It is caused by the constant collision of lighter fluid molecules with heavier particles.
3. Who discovered Brownian motion?
Answer: Brownian motion was discovered by Scottish botanist Robert Brown in 1827 while observing pollen particles in water under a microscope.
4. What causes Brownian motion?
Answer: Brownian motion is caused by the collision of lighter fluid molecules (like water or air) with heavier particles (such as pollen or smoke), resulting in random movement.
5. What is diffusion?
Answer: Diffusion is the process by which particles spread from an area of high concentration to an area of low concentration until they are evenly distributed.
6. How does diffusion occur?
Answer: Diffusion occurs due to the random motion of particles, causing them to move and collide, spreading from regions of higher concentration to regions of lower concentration.
7. What factors affect the rate of diffusion?
Answer: Factors affecting the rate of diffusion include temperature, particle size, the concentration gradient, and the medium through which diffusion occurs.
8. How does temperature affect particle movement?
Answer: Higher temperatures increase the kinetic energy of particles, causing them to move faster and collide more frequently. This increases the rate of diffusion and pressure in gases.
9. What is kinetic energy in the context of particle movement?
Answer: Kinetic energy is the energy possessed by particles due to their motion. Higher temperatures result in higher kinetic energy, leading to faster particle movement.
10. How does particle movement differ in solids, liquids, and gases?
Answer: In solids, particles vibrate in fixed positions. In liquids, particles move more freely but remain close together. In gases, particles move rapidly and are far apart.
11. What is the relationship between temperature and pressure in gases?
Answer: In gases, an increase in temperature causes particles to move faster, leading to more frequent and forceful collisions with container walls, increasing pressure.
12. What is thermal expansion?
Answer: Thermal expansion is the increase in volume of a substance due to an increase in temperature, which causes particles to move faster and occupy more space.
13. How does particle size affect diffusion?
Answer: Smaller particles diffuse faster than larger particles because they move more easily and are less affected by obstacles in their path.
14. What is the concentration gradient in diffusion?
Answer: The concentration gradient is the difference in concentration of particles between two regions. The steeper the gradient, the faster the rate of diffusion.
15. How is Brownian motion evidence of the kinetic theory of matter?
Answer: Brownian motion provides evidence for the kinetic theory of matter, which states that particles are in constant, random motion and that this motion increases with temperature.
16. What is meant by random particle movement?
Answer: Random particle movement refers to the unpredictable and irregular motion of particles, as seen in Brownian motion and diffusion.
17. How does particle movement contribute to gas pressure?
Answer: In gases, particles move rapidly and collide with container walls. The force exerted by these collisions creates pressure.
18. What is an example of diffusion in everyday life?
Answer: An example of diffusion is the spreading of perfume in a room. The perfume particles move from an area of high concentration (near the bottle) to low concentration (throughout the room).
19. How does the medium affect the rate of diffusion?
Answer: The rate of diffusion varies depending on the medium. Diffusion is faster in gases, slower in liquids, and slowest in solids due to the differences in particle mobility.
20. What is the role of temperature in phase changes?
Answer: Temperature affects phase changes by altering particle movement. Increasing temperature can cause melting and boiling, while decreasing temperature can cause freezing and condensation.
21. How does diffusion help in biological processes?
Answer: Diffusion is crucial in biological processes such as gas exchange in the lungs, nutrient absorption in the intestines, and the movement of ions across cell membranes.
22. What is the difference between diffusion and osmosis?
Answer: Diffusion is the movement of particles from high to low concentration, while osmosis is the movement of water molecules through a semipermeable membrane from low to high solute concentration.
23. What is the significance of Brownian motion in chemistry?
Answer: Brownian motion is significant in chemistry as it provides evidence for the existence and constant motion of molecules, supporting the kinetic theory of matter.
24. What is the effect of particle collisions on diffusion?
Answer: Particle collisions cause particles to spread out and mix, leading to diffusion. The frequency and intensity of collisions increase with temperature, speeding up diffusion.
25. How is diffusion different from convection?
Answer: Diffusion is the random movement of particles spreading out, while convection involves the bulk movement of a fluid, carrying particles with it, typically due to temperature differences.
26. How does surface area affect the rate of diffusion?
Answer: A larger surface area allows more particles to diffuse at once, increasing the rate of diffusion.
27. What is the role of diffusion in respiration?
Answer: Diffusion allows oxygen to move from the alveoli in the lungs into the blood and carbon dioxide to move from the blood into the alveoli for exhalation.
28. How do cells use diffusion to obtain nutrients?
Answer: Cells use diffusion to obtain nutrients by allowing dissolved substances to move from areas of high concentration outside the cell to lower concentration inside the cell.
29. What is the difference between simple and facilitated diffusion?
Answer: Simple diffusion occurs without assistance, while facilitated diffusion requires transport proteins to help move particles across cell membranes.
30. How does the diffusion of gases differ from the diffusion of liquids?
Answer: Gases diffuse faster than liquids because gas particles move more freely and have greater kinetic energy compared to liquid particles.
31. What is the effect of particle movement on the state of matter?
Answer: Particle movement determines the state of matter. Increased movement can change solids to liquids (melting) and liquids to gases (boiling), while decreased movement can change gases to liquids (condensation) and liquids to solids (freezing).
32. What is dynamic equilibrium in the context of diffusion?
Answer: Dynamic equilibrium is reached when particles continue to move but there is no net change in concentration because the particles are evenly distributed.
33. How do particle interactions lead to Brownian motion?
Answer: Brownian motion results from the constant and random collisions between lighter fluid molecules and heavier suspended particles, causing the particles to move erratically.
34. What is the significance of Brownian motion in the study of particles?
Answer: Brownian motion provides direct evidence of the existence and constant motion of particles, supporting the kinetic theory and molecular theory of matter.
35. What is the impact of particle movement on pressure in a closed system?
Answer: In a closed system, increased particle movement due to higher temperature leads to more frequent and forceful collisions with container walls, increasing pressure.
36. How does particle movement explain the behavior of gases?
Answer: Particle movement explains the behavior of gases, including expansion to fill containers, diffusion, pressure, and temperature effects, as particles move rapidly and freely.
37. How is Brownian motion observed in a laboratory setting?
Answer: Brownian motion is observed by suspending tiny particles like pollen or smoke in a fluid and viewing their erratic movement under a microscope.
38. What is the role of particle movement in chemical reactions?
Answer: Particle movement facilitates chemical reactions by increasing the frequency and energy of collisions between reactant molecules, leading to the formation of new products.
39. How does particle movement affect the viscosity of liquids?
Answer: Higher particle movement (due to increased temperature) decreases the viscosity of liquids, making them flow more easily, while lower movement increases viscosity.
40. What is the relationship between particle movement and thermal energy?
Answer: Particle movement is directly related to thermal energy; as thermal energy increases, particles move faster and have greater kinetic energy.
41. How do temperature changes affect the diffusion rate of particles?
Answer: Increasing temperature increases the kinetic energy of particles, speeding up diffusion. Decreasing temperature slows down particle movement, reducing the diffusion rate.
42. How does the medium of diffusion impact particle movement?
Answer: The medium impacts particle movement; diffusion is faster in gases due to low resistance, slower in liquids, and slowest in solids due to higher resistance and particle density.
43. What is the effect of concentration gradients on particle movement?
Answer: Particles move from high to low concentration areas. The steeper the concentration gradient, the faster the rate of diffusion until equilibrium is reached.
44. How does particle movement relate to the concept of entropy?
Answer: Particle movement increases entropy, as particles spread out and mix more randomly, leading to greater disorder and energy dispersal.
45. What is the significance of particle movement in the states of matter?
Answer: Particle movement determines the properties of solids, liquids, and gases. In solids, movement is restricted; in liquids, it is freer; in gases, particles move rapidly and independently.
46. How do intermolecular forces affect particle movement?
Answer: Strong intermolecular forces restrict particle movement, as seen in solids and liquids, while weak forces allow more freedom of movement, as seen in gases.
47. How is particle movement measured?
Answer: Particle movement can be measured using techniques like Brownian motion analysis, diffusion coefficients, and temperature changes affecting kinetic energy.
48. How does particle movement explain the behavior of fluids?
Answer: Particle movement in fluids explains properties like viscosity, flow, diffusion, and pressure changes, as particles move and interact within the fluid.
49. What is the effect of particle movement on the physical properties of materials?
Answer: Particle movement affects physical properties like temperature, pressure, viscosity, and phase changes, influencing how materials behave under different conditions.
50. How does particle movement impact the rate of reaction in chemistry?
Answer: Particle movement impacts the rate of reaction by affecting the frequency and energy of collisions between reactant molecules, with higher movement generally leading to faster reactions.
